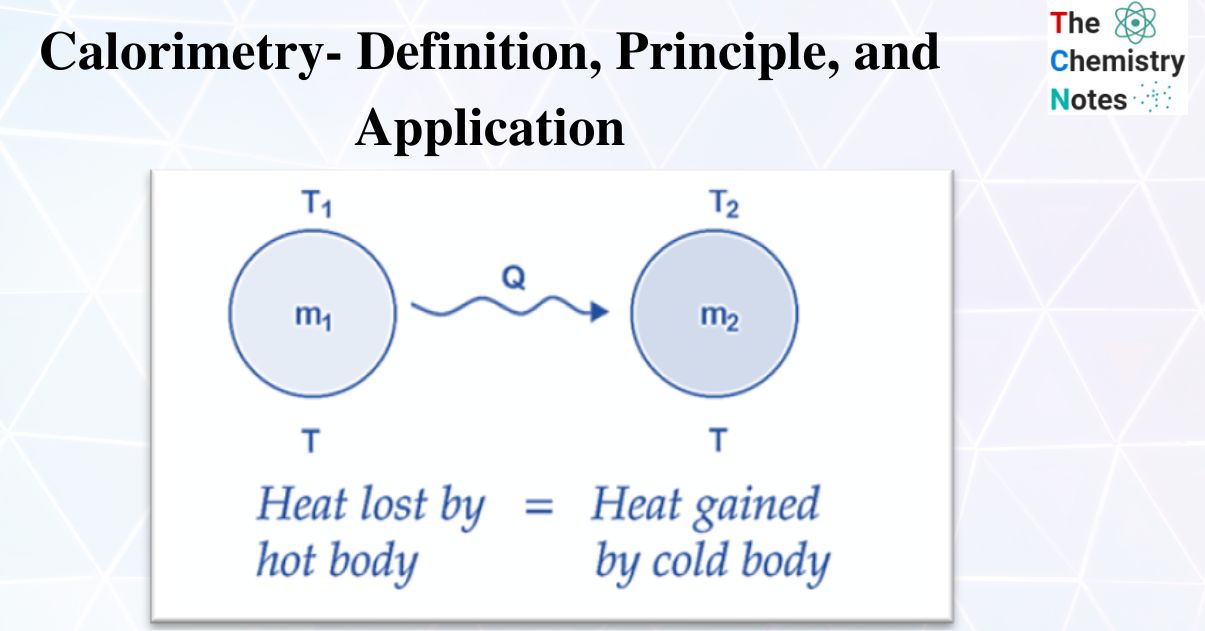
Energy Calorimetry Definition .calorimetry is used to measure amounts of heat transferred to or from a substance. To do so, the heat is exchanged with a.
from thechemistrynotes.com
in chemistry and thermodynamics, calorimetry (from latin calor 'heat', and greek μέτρον (metron) 'measure') is the.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. For example, when an exothermic reaction.
Calorimetry Definition, Principle, Types, Application, and Limitations
Energy Calorimetry Definition a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.calorimetry is used to measure amounts of heat transferred to or from a substance.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.
From schoolbag.info
Heat Thermochemistry Training MCAT General Chemistry Review Energy Calorimetry Definitionupdated on february 21, 2019.calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction.calorimetry is used to measure amounts of heat transferred to or from a substance.thermal energy itself cannot be measured easily, but the temperature change caused by the flow of thermal energy between. Web. Energy Calorimetry Definition.
From www.tec-science.com
Calorimeter to determine the specific heat capacities of liquids tec Energy Calorimetry Definitioncalorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction.calorimetry is used to measure amounts of heat transferred to or from a substance. Calorimetry is a method of measuring the heat transfer within a.updated on february 21, 2019. For example, when an exothermic reaction. Energy Calorimetry Definition.
From martinfersbanks.blogspot.com
Is a Bomb Calorimeter Constant Pressure Energy Calorimetry Definitioncalorimetry is used to measure amounts of heat transferred to or from a substance.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimetry is a method of. Energy Calorimetry Definition.
From www.animalia-life.club
Calorimeter Diagram Energy Calorimetry Definitioncalorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.updated on february 21, 2019. Calorimetry is a method of measuring the heat transfer within a.a container that prevents. Energy Calorimetry Definition.
From www.youtube.com
Thermal Properties of Matter Class 11 Physics Calorimetry Principle Energy Calorimetry Definition Calorimetry is a method of measuring the heat transfer within a.thermal energy itself cannot be measured easily, but the temperature change caused by the flow of thermal energy between.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.calorimetry is the process of measuring the amount. Energy Calorimetry Definition.
From www.youtube.com
Calorimetry Lecture 2 Thermal Physics Water Equivalent of Substance Law Energy Calorimetry Definitionthermal energy itself cannot be measured easily, but the temperature change caused by the flow of thermal energy between. To do so, the heat is exchanged with a.calorimetry is used to measure amounts of heat transferred to or from a substance.a calorimeter is a device used to measure the amount of heat involved in a. Energy Calorimetry Definition.
From thechemistrynotes.com
Calorimetry Definition, Principle, Types, Application, and Limitations Energy Calorimetry Definitionupdated on february 21, 2019. To do so, the heat is exchanged with a.a container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements.calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. Calorimetry is a method. Energy Calorimetry Definition.
From engineeringlearn.com
Bomb Calorimeter Definition, Construction, Diagram, Working & Uses Energy Calorimetry Definitiona calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. For example, when an exothermic reaction.thermal energy itself cannot be measured easily, but the temperature change caused by the flow. Energy Calorimetry Definition.
From socratic.org
Finding DeltaH given heat released and mass in bomb calorimetry? Socratic Energy Calorimetry Definitiona calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.updated on february 21, 2019.thermal energy itself cannot be measured easily, but the temperature change caused by the flow of thermal energy between.in chemistry and thermodynamics, calorimetry (from latin calor 'heat', and greek μέτρον (metron). Energy Calorimetry Definition.
From www.youtube.com
Calorimetry Graphs GCSE Science grade 7, 8 and 9 Booster Questions Energy Calorimetry Definition For example, when an exothermic reaction.calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction.a container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements.updated on february 21, 2019.in chemistry and thermodynamics, calorimetry (from. Energy Calorimetry Definition.
From www.pathwaystochemistry.com
Calorimetry Pathways to Chemistry Energy Calorimetry Definitiona container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.calorimetry is used to measure amounts of heat transferred to or from a substance.updated. Energy Calorimetry Definition.
From www.slideshare.net
Energy Energy Calorimetry Definitionupdated on february 21, 2019.a container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. Calorimetry is a method of measuring the heat transfer within a.. Energy Calorimetry Definition.
From www.youtube.com
050 Calorimetry YouTube Energy Calorimetry Definitionin chemistry and thermodynamics, calorimetry (from latin calor 'heat', and greek μέτρον (metron) 'measure') is the.calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction.a container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements.a. Energy Calorimetry Definition.
From pressbooks.online.ucf.edu
10.2 Calorimetry Chemistry Fundamentals Energy Calorimetry Definitiona container that prevents heat transfer in or out is called a calorimeter, and the use of a calorimeter to make measurements.updated on february 21, 2019. For example, when an exothermic reaction.thermal energy itself cannot be measured easily, but the temperature change caused by the flow of thermal energy between. To do so, the heat. Energy Calorimetry Definition.
From gamesmartz.com
Calorimeter Definition Easy to Understand Energy Calorimetry Definitioncalorimetry is used to measure amounts of heat transferred to or from a substance. Calorimetry is a method of measuring the heat transfer within a.thermal energy itself cannot be measured easily, but the temperature change caused by the flow of thermal energy between. To do so, the heat is exchanged with a.in chemistry and thermodynamics,. Energy Calorimetry Definition.
From rainis.pics
Calorimeter Types and Heat Flow Analysis (M6Q5) UWMadison Chemistry Energy Calorimetry Definitionthermal energy itself cannot be measured easily, but the temperature change caused by the flow of thermal energy between.updated on february 21, 2019.calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction. For example, when an exothermic reaction.in chemistry and thermodynamics, calorimetry (from latin calor 'heat',. Energy Calorimetry Definition.
From study.com
What is a Calorimeter? Definition, Uses & Equation Video & Lesson Energy Calorimetry Definitioncalorimetry is used to measure amounts of heat transferred to or from a substance.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process.in chemistry and thermodynamics, calorimetry (from latin calor 'heat', and greek μέτρον (metron) 'measure') is the. To do so, the heat is exchanged with. Energy Calorimetry Definition.
From www.youtube.com
Principle of Calorimetry YouTube Energy Calorimetry Definitioncalorimetry is used to measure amounts of heat transferred to or from a substance.a calorimeter is a device used to measure the amount of heat involved in a chemical or physical process. To do so, the heat is exchanged with a.a container that prevents heat transfer in or out is called a calorimeter, and the. Energy Calorimetry Definition.